|
We need to calculate the change in entropy of both system and surroundings and then add them to get the change in entropy of the Universe.įirst of all we know that this process is completely irreversible therefore $dS \neq \frac \neq 0$, even though the process has been reversible and therefore the Universe must undergo no change in entropy. The lake is a heat bath and the water added is our small thermodynamic system. This is a homework question but I believe it touches on something very general. What is the change in entropy of the Universe? So if, say, you have an enthalpy change of -92.2 kJ mol-1, the value you must put into the equation is -92200 J mol-1.Water at temperature $T_s$ is added to a lake at temperature $T_0 > T_s$ until the total system reaches equilibrium. That means that if you are calculating entropy change, you must multiply the enthalpy change value by 1000. The total entropy change is simply the sum of the system and the surroundings. If we have the entropy changes of the system and surroundings, we can calculate total entropy change. How do you calculate entropy in chemistry? What is total entropy? If you notice, we dont have an expression for the way entropy changes due to. 
When we hold temperature constant (an isothermal process), and change one of the other parameters: (1) S P 2 P 1 ( S P)T dP. What do u mean by entropy?Įntropy is a scientific concept as well as a measurable physical property that is most commonly associated with a state of disorder, randomness, or uncertainty. Entropy is typically considered a function of temperature and either volume or pressure. Generally, entropy is defined as a measure of randomness or disorder of a system. The solid wood burns and becomes ash, smoke and gases, all of which spread energy outwards more easily than the solid fuel. The addition of a sodium ion to a chloride ion to form sodium chloride is an example of a reaction you can calculate this way. If you know these quantities, use the following formula to work out the overall change: H Hproducts Hreactants. We see evidence that the universe tends toward highest entropy many places in our lives. The most basic way to calculate enthalpy change uses the enthalpy of the products and the reactants. 1 What is entropy with example?Įntropy is a measure of the energy dispersal in the system. What is Delta S Equation? Change in entropy (delta S), at constant temperature is defined as: ΔS=ΔQT Δ S = Δ Q T. Thus, for a system at equilibrium, ΔG=0, and then we find that ΔS=ΔHT. Therefore, the free energy expression provides a relationship between enthalpy and entropy. 
where at constant temperature, the change on free energy is defined as: ΔG=ΔH−TΔS. To calculate the entropy change in Excel follow the steps below. The concept of entropy provides deep insight into the direction of spontaneous change for many everyday phenomena.2 How do you calculate entropy and enthalpy?Įntropy ( S ) defines the degree of randomness or disorder in a system. The entropy change formula is (xijlog2 (xij)). Why do we calculate entropy?īecause work is obtained from ordered molecular motion, the amount of entropy is also a measure of the molecular disorder, or randomness, of a system. Since each reservoir undergoes an internally reversible, isothermal process, the entropy change for each reservoir can be determined from ΔS = Q/T where T is the constant absolute temperature of the system and Q is the heat transfer for the internally reversible process. The absolute temperature is denoted by the letter T. 
Select cell A and move cursor to the bottom right of the cell, a black plus sign will appear, drag the plus sign to cell C5 to copy the formula in these cell. Click on Cell A5 and write this formula SUM (A2:A4). 29.2 x calculate the increase of entropy (in j/k) when 68 g of ice melts at 0 °c and 1 atm. Before calculating the entropy change you first have to find out the sum of each series. 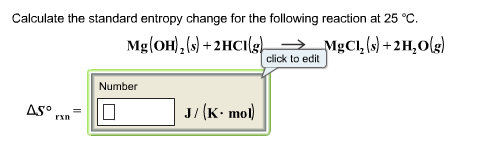
Using the change in entropy formula, we can compute the Entropy Change of a chemical reaction or a system: S (Q/T)rev Where, The heat transfer to or from the thermodynamic system is denoted by Q. Calculate the entropy change associated with evaporating one glass (250 g) of sweat by a human body at 298 k and 1 bar thermo refers to heat b on the other, a change in. # entropy(q) = -( q * log2(q) + (1-q) * log2(1-q) )Įntropy = -(q * np.log2(q) + (1-q) * np.log2(1-q))īy way of numerous illustrations, we have demonstrated how to use code written to solve the Calculate Entropy problem. A thermodynamic system’s Entropy Change is denoted by the letter S.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
